Procedure
|
Elements
Na-Sodium Mg-Magnesium Al-Aluminum Si-Silicon P-Phosphorus S-Sulfur Cl-Chlorine Ar-Argon |
Properties
Atomic number 11 Atomic mass 22.98977 g/mol Density 0.97 g.cm -3 at 20 °C soft metal with low melting point. Atomic Number 12 Atomic mass 24.30 g/mol silvery white and light with density of 1740 kg/m^3 Atomic Number 13 Atomic mass 26.98 g/mol soft, lightweight metal with a dull silvery appearance. Atomic number 14 Atomic mass 28.09 g/mol A metalloid with a marked metallic lust. Very brittle. Atomic number 14 Atomic mass 30.97 g/mol A multivalent nonmental in group 15. Found in several allotropic forms. (same as above) Atomic number 16 Atomic mass 32.06 g/mol A multivalent non-metal, tasteless, odorless, found in its native form as a yellow crystalline solid. Atomic number 17 Atomic mass 35.45 g/mol A halogen. Chlorine gas is greenish-yellow and combines with nearly all other elements. Atomic Number 18 Atomic mass 39.95 g/mol A noble gas, soluble in water, colorless and odorless in both liquid and gaseous states. |
Reactivity
Most Reactive metal because it's an Alkali metal. 2nd most reactive comparing to sodium. It's in the group Alkaline Earth metal whichis the second most reactive metals. not very good chemical reactivity Reacts with all halogens to form silicon tetra halides. A chemical reactivity of phosphorus only occurs in nature combined with oxygen or other elements in the form of phosphates. 2nd most reactive non-metal. However, at normal temperatures, sulfur is non-reactive. Most reactive non-metals because it only needs to gain one electron to become stable. Does not react because it has eight valence electrons. (already stable) |
Analysis and Conclusions
1. The atomic radius of a chemical element is the measure of the size of its atom. This is typically the distance from the nucleus to the boundary of the surrounding cloud of electrons.
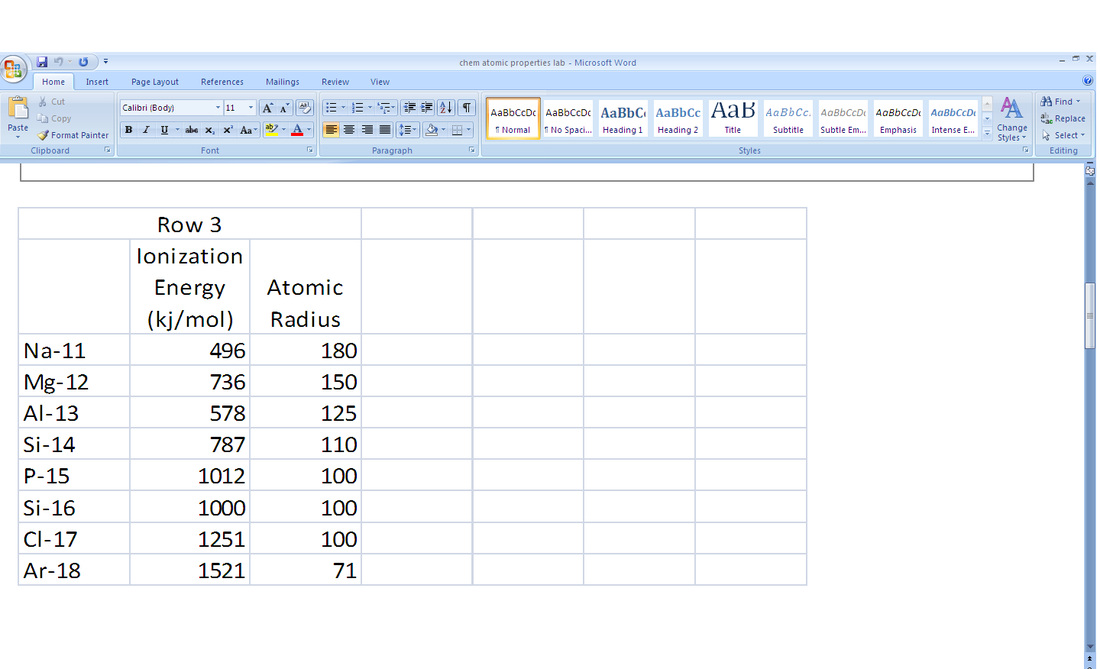
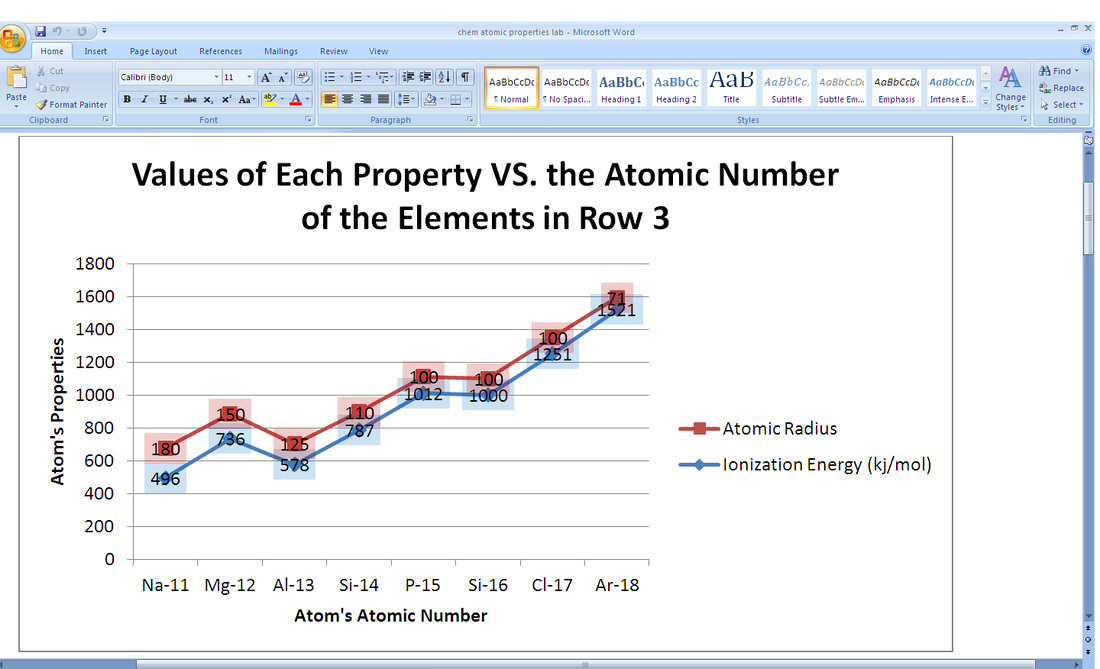
2. According to the graphs above, the trends in atomic radius increases as you move left and down the periodic table. For example, row three, starting at Argon, one will notice the element has a particularly small atomic radius. As one moves to the element on the left (Chloride), the atomic radius expands and gets larger. This goes the same when one is moving down the periodic table's groups.
3.The factors that determine these trends in the atomic radius are that it increases going down the groups because of the increasing energy levels. It also decreases going right across the rows of the periodic table because of the increasing effective nuclear charge due to decreased shielding. (It has more protons which sucks electrons.)
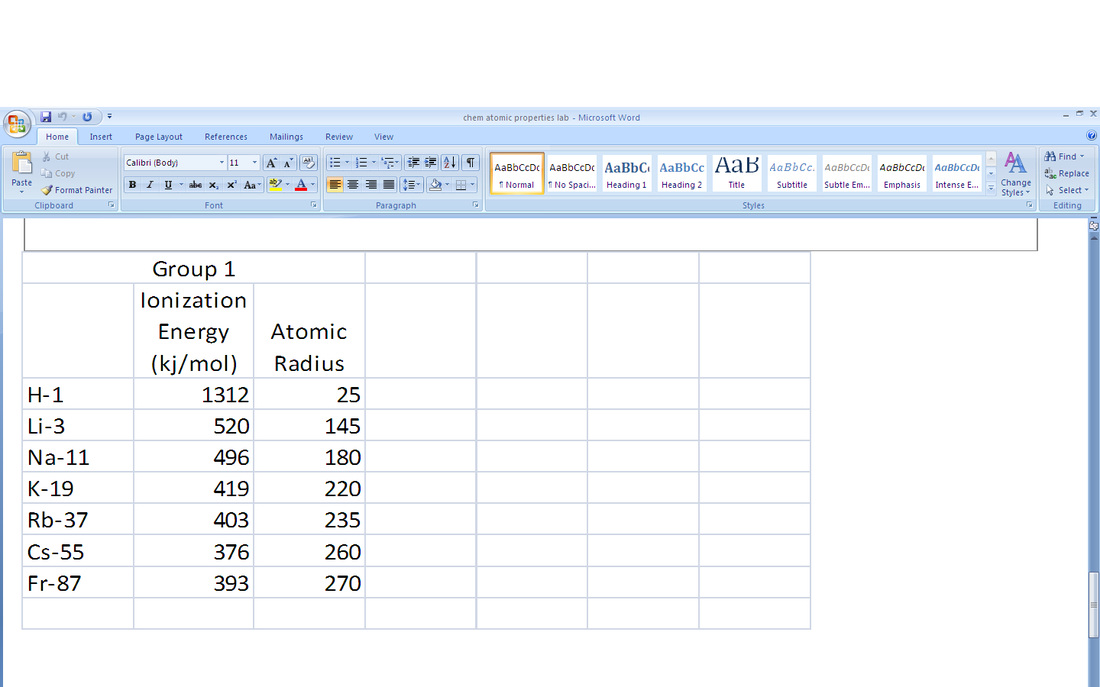
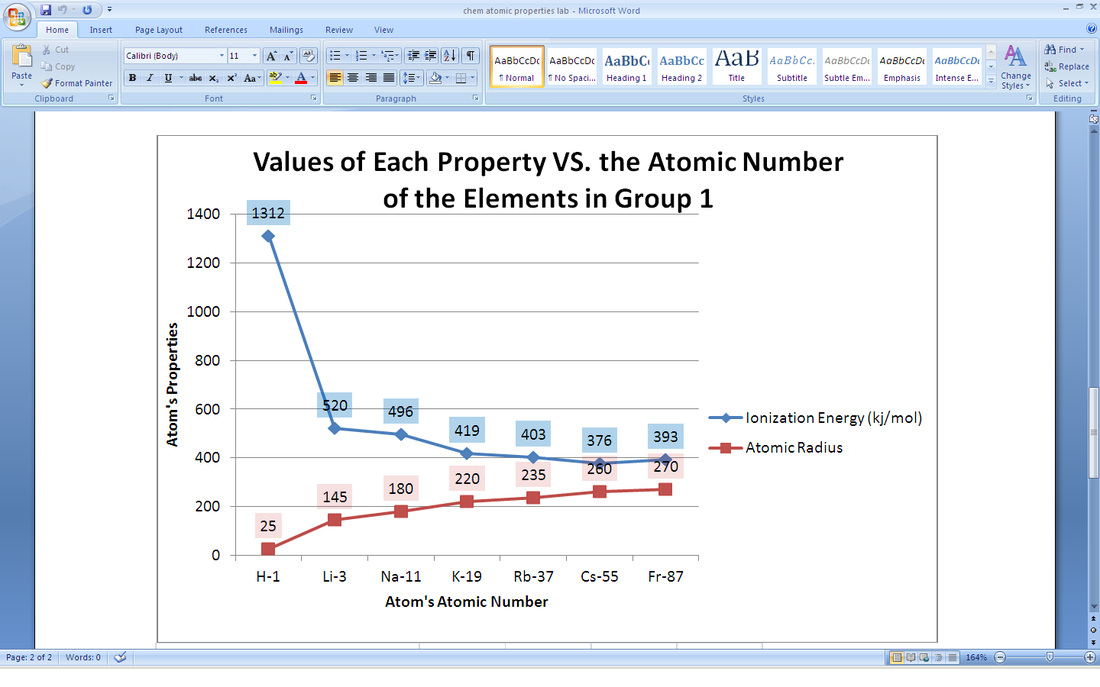
4. The trend in ionic radii of period 3 and group 1 were total opposites. The ionic radius would increase up and right on the periodic table while the atomic radius would increase left and down the periodic table. As more protons are added, it takes more energy to pull off an electron. Going down, according to the shielding effect, it takes less energy to pull off an electron.
5. The first ionization energy is the energy it takes to remove an electron from an atom.
6. According to the graphs above, the trends in the ionization energy increases as you move up and right on the periodic table. For example, row three, starting at Sodium (Na), one will notice the element has a particularly small ionization energy. As one moves to the element on the right (Mg), the ionization energy increases. This goes the same when one is moving up the periodic table's groups.
7. The factors that determine these trends in the atomic radius are that it increases going across the periodic to the right. This is because electrons in the same quantum level do not shield as effectively as electrons in inner levels and the increasing nuclear charge. Ionization energy increases for successive electrons. The line in the graph isn't smooth as one moves across the row, however, because there are irregularities within the rows at half-filled and filled sublevels, due to extra repulsion of electrons paired in orbitals, making them easier to remove. Ionization energy decreases with increasing atomic number within a group (going down periodic table). This is because electrons farther from the nucleus are easier to remove. (It increases going up the periodic table because of the shielding effect.)
8.Electron affinity is the attraction a non-bonded atom has for an electron. Electron affinity increases up and to the right just like the ionization energy. Affinity tends to decrease as you go left in a period because electrons farther from the nucleus experience a less nuclear attraction. Adding on to that, there are also some irregularites due to repulsive forces in the small p orbitals.
9. The characteristics of a metal are that they are good conductors of heat and electricity, malleable, ductile, shiny, and there’s few electrons in the outer energy level. The characteristics of a nonmetal are that it has almost a complete set of electrons in the outer energy level, it’s not a good conductor of heat and electricity, it’s not malleable, it’s not ductile, and it’s not shiny. The characteristics of a metalloid are that it has half a complete set of electrons, they’re also known as semiconductors and it has the same properties of metals and nonmetals although the density, melting/boiling points may vary. The characteristics of a noble gas are that it has a complete set of electrons in the outer energy level, they’re gases at room temperature, they’re nonmetals, colorless, odorless, found in small amounts and are not reactive.
10. Na: metal
Mg: metal
Al: metal
Si: metalloids
P: nonmetal
Cl: nonmetal
Ar: noble gas
11. Group 1 and group 17 are the most active metals and nonmetals because it takes them less energy to lose or gain one electron to become stable as a noble gas. All atoms try to have eight valence electrons to become stable. Group 1 can easily get rid of an electron to become stable. Group 17 already has seven electrons, so it can take one electron from another atom to become stable.
2. According to the graphs above, the trends in atomic radius increases as you move left and down the periodic table. For example, row three, starting at Argon, one will notice the element has a particularly small atomic radius. As one moves to the element on the left (Chloride), the atomic radius expands and gets larger. This goes the same when one is moving down the periodic table's groups.
3.The factors that determine these trends in the atomic radius are that it increases going down the groups because of the increasing energy levels. It also decreases going right across the rows of the periodic table because of the increasing effective nuclear charge due to decreased shielding. (It has more protons which sucks electrons.)
4. The trend in ionic radii of period 3 and group 1 were total opposites. The ionic radius would increase up and right on the periodic table while the atomic radius would increase left and down the periodic table. As more protons are added, it takes more energy to pull off an electron. Going down, according to the shielding effect, it takes less energy to pull off an electron.
5. The first ionization energy is the energy it takes to remove an electron from an atom.
6. According to the graphs above, the trends in the ionization energy increases as you move up and right on the periodic table. For example, row three, starting at Sodium (Na), one will notice the element has a particularly small ionization energy. As one moves to the element on the right (Mg), the ionization energy increases. This goes the same when one is moving up the periodic table's groups.
7. The factors that determine these trends in the atomic radius are that it increases going across the periodic to the right. This is because electrons in the same quantum level do not shield as effectively as electrons in inner levels and the increasing nuclear charge. Ionization energy increases for successive electrons. The line in the graph isn't smooth as one moves across the row, however, because there are irregularities within the rows at half-filled and filled sublevels, due to extra repulsion of electrons paired in orbitals, making them easier to remove. Ionization energy decreases with increasing atomic number within a group (going down periodic table). This is because electrons farther from the nucleus are easier to remove. (It increases going up the periodic table because of the shielding effect.)
8.Electron affinity is the attraction a non-bonded atom has for an electron. Electron affinity increases up and to the right just like the ionization energy. Affinity tends to decrease as you go left in a period because electrons farther from the nucleus experience a less nuclear attraction. Adding on to that, there are also some irregularites due to repulsive forces in the small p orbitals.
9. The characteristics of a metal are that they are good conductors of heat and electricity, malleable, ductile, shiny, and there’s few electrons in the outer energy level. The characteristics of a nonmetal are that it has almost a complete set of electrons in the outer energy level, it’s not a good conductor of heat and electricity, it’s not malleable, it’s not ductile, and it’s not shiny. The characteristics of a metalloid are that it has half a complete set of electrons, they’re also known as semiconductors and it has the same properties of metals and nonmetals although the density, melting/boiling points may vary. The characteristics of a noble gas are that it has a complete set of electrons in the outer energy level, they’re gases at room temperature, they’re nonmetals, colorless, odorless, found in small amounts and are not reactive.
10. Na: metal
Mg: metal
Al: metal
Si: metalloids
P: nonmetal
Cl: nonmetal
Ar: noble gas
11. Group 1 and group 17 are the most active metals and nonmetals because it takes them less energy to lose or gain one electron to become stable as a noble gas. All atoms try to have eight valence electrons to become stable. Group 1 can easily get rid of an electron to become stable. Group 17 already has seven electrons, so it can take one electron from another atom to become stable.
Application of Ideas
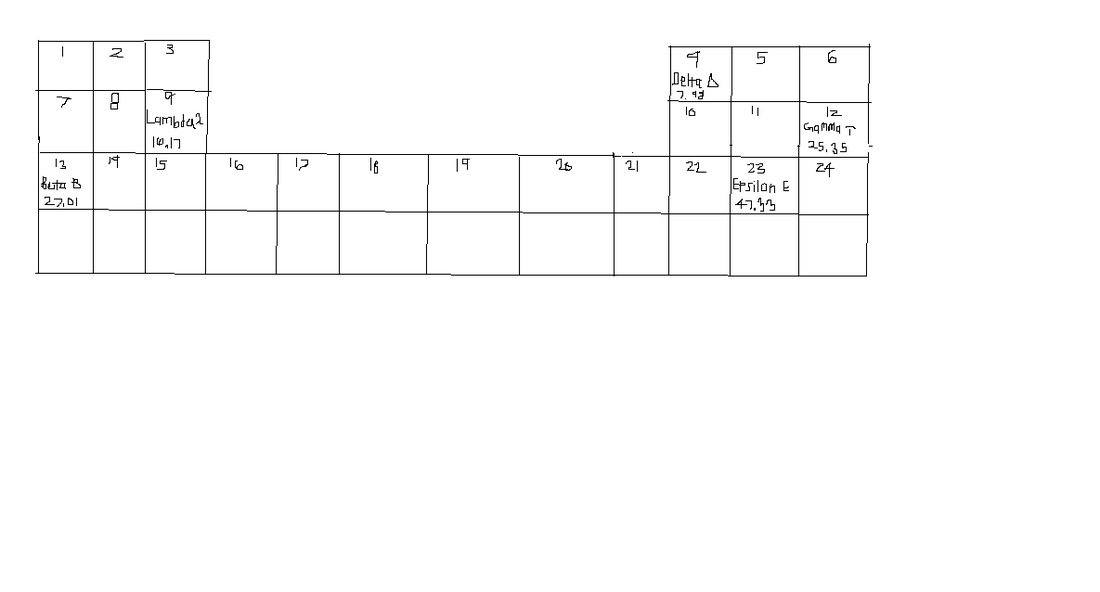
2. Based off the periodic table created, an element with an atomic mass of 11.29 that has nonmetallic propeties and is very reactive would have the atomic number of 5.
3. Based off the periodic table created, an element with an atomic mas of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta would have an atomic number of 8.
4. The elements towards the left are metals, whilst the elements on the right are nonmetal. The atomic number increases as it goes left to right across the periodic table. The group Gamma is in would be unreactive and stable like noble gases. The group Epsilon and Beta are in would be the most reactive groups.
5. Out of these elements, the one with the largest atomic radius would be Beta. The one with the smallest atomic radius would be Delta. Out of these elements, the one with the highest first ionization enery would be Delta . The lowest ionization energy would be Beta.
3. Based off the periodic table created, an element with an atomic mas of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta would have an atomic number of 8.
4. The elements towards the left are metals, whilst the elements on the right are nonmetal. The atomic number increases as it goes left to right across the periodic table. The group Gamma is in would be unreactive and stable like noble gases. The group Epsilon and Beta are in would be the most reactive groups.
5. Out of these elements, the one with the largest atomic radius would be Beta. The one with the smallest atomic radius would be Delta. Out of these elements, the one with the highest first ionization enery would be Delta . The lowest ionization energy would be Beta.