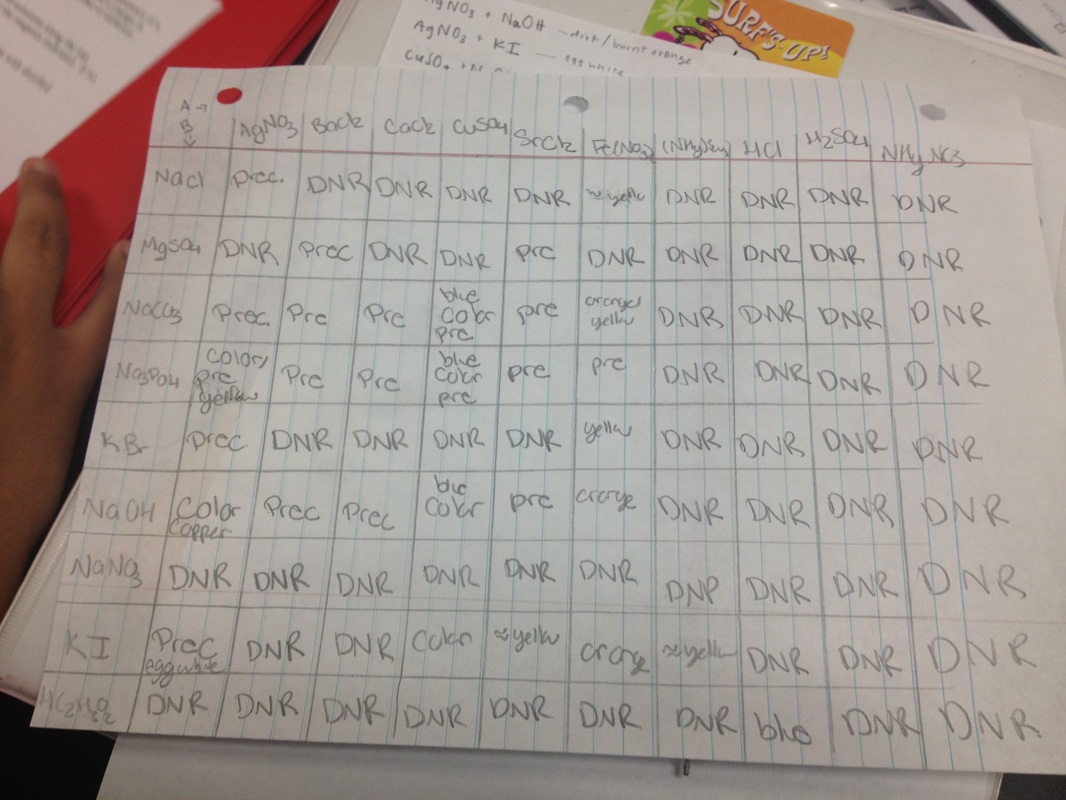
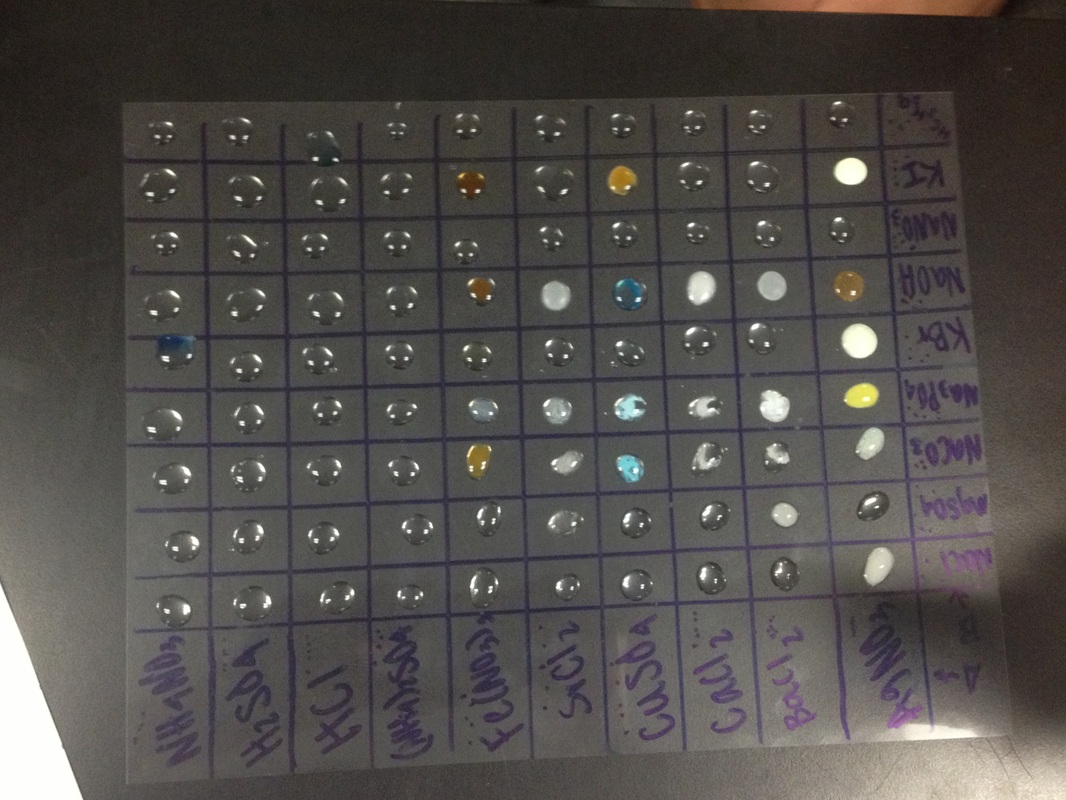
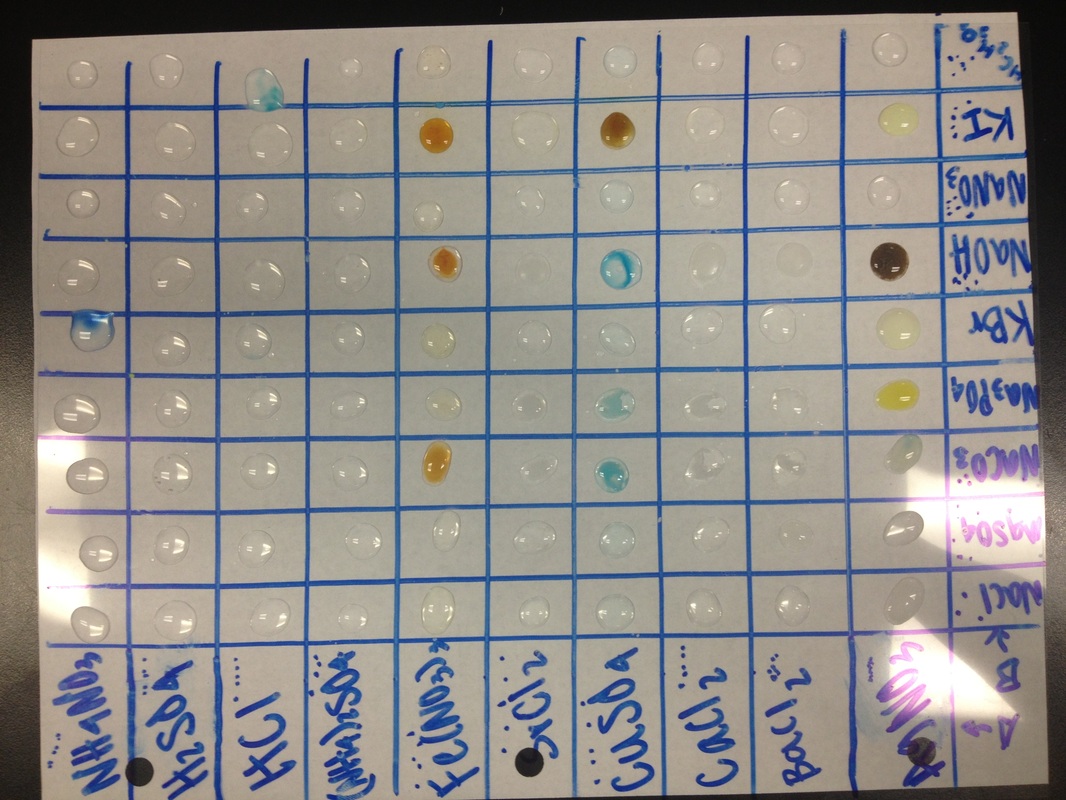
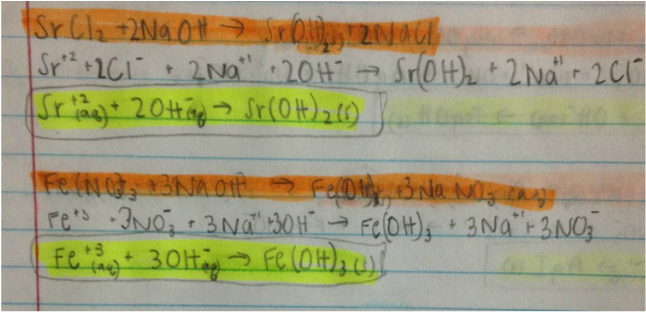Data Table
Conclusion
A group of selected solutions (group A) were mixed together with another group of solutions (group B) to be observed for physical changes. The different substances combined gave a variety of reactions such as the appearance of a solid precipitate, a change in color of a solution or a generation of a gas.
Discussion of Theory
This lab required mixing solutions together and observing physical changes. Some had no reactions, some had color changes, and some produced a little precipitate. The double replacement reactions that had two products come out as aqueous showed no reaction. There are three types of metathesis reactions. They are precipitate reaction, neutralization reaction, and gas formation reaction. Although a gas formation reaction was not present in this lab, the other two types of metathesis reactions were. In order to find out if a balanced equation were to have a precipitate, a net ionic equation was written. A net ionic equation includes only those components that take part in the chemical change. Thus, the spectator ions are eliminated because they "sit and do nothing". After writting out the molecular equation, one had to remember the solubility rules. A product is soluble (aqueous) if it dissolves in water. The solute is what gets dissolved and the solvent is what does the dissolving. Using the solubility rules, one can determine if the products are aqueous, solid, liquid or gas in the molecular equation and can then proceed to writing out the net ionic equation. Neutralized reactions tend to have H2O in the products.
Sources of Error
The few errors that may have occurred are most likely random errors. The procedure of dropping the solutions was not definite. There were times when the miniature scientists dropped two drops of solution A and one drop of solution B. This may have caused a disturbance in the reaction which would then cause an inaccurate reading of the reaction. A few times, due to the lighting, it might have been hard to tell if there was a reaction. There were no systematic errors that were obvious that could've askewed the data collected.
Analysis Questions
1. Some specific cations and anions left a reaction. AgNO3 + NaOH left a dirt/burnt orange color reaction. AgNO3 + KI left an egg white color. Fe(NO3)3 + Na2PO4 left an amber color.
3. A metathesis reaction is a reaction where the cations and anions exhchange partners. There are three types are precipitate reaction, neutralization reaction and gas formation reaction. The precipitate reaction is when the products form a solid which we call precipitates. An example of one would be Fe(NO3)3 (aq) + 3Na(OH)(aq) --> Fe(OH)3 (s)+ 3NaNO3 (aq). The neutralization reaction would be when the products formed are water molecules. An example would be HCl(aq) + Na(OH) (aq) --> H2O (l) + NaCl (aq). It's not often when a gas forms, and it certainly did not occur in this lab. An example of of one, however, would be 2H+(aq) + CO3(-2 charge) (aq) --> H2O(l) + CO2 (g)
4. Some things are soluble in water while others are not because it has to create a homogenous solution. The solubility of a solute depends on the type of solvent, the temperature and the pressure. Some things are very soluble and some are just kind of soluble -meaning they aren’t completely soluble and may need some assistance such as more heat. A helpful hint is “like dissolves like”. This means water dissolves water soluble things and oil dissolves oil soluble things. Water is polar while oil is non-polar. Water dissolves nonionic substances.
5. A double replacement reaction that produces two aqueous products is considered a "no reaction" because it indicates that all the subsances are completely soluble. Thus, if you wrote out the net ionic equation, everything (ions and spectator ions) would cancel out leaving you left with a "no reaction."
4. Some things are soluble in water while others are not because it has to create a homogenous solution. The solubility of a solute depends on the type of solvent, the temperature and the pressure. Some things are very soluble and some are just kind of soluble -meaning they aren’t completely soluble and may need some assistance such as more heat. A helpful hint is “like dissolves like”. This means water dissolves water soluble things and oil dissolves oil soluble things. Water is polar while oil is non-polar. Water dissolves nonionic substances.
5. A double replacement reaction that produces two aqueous products is considered a "no reaction" because it indicates that all the subsances are completely soluble. Thus, if you wrote out the net ionic equation, everything (ions and spectator ions) would cancel out leaving you left with a "no reaction."